PACEMAKER OR DEFIBRILLATOR (ICD) FOLLOW-UP 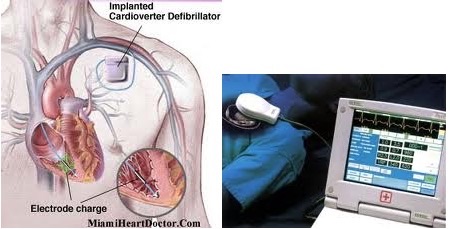
Regular follow-up checks of your pacemaker or implantable cardioverter-defibrillator (ICD) are an important part of your implanted device care. Some of the objectives of the pacemaker follow-up clinic include:
- 1) optimize the settings of the pacemaker to match your present needs,
- 2) maximize the battery life of your pacemaker and detect early signs of battery depletion,
- 3) monitor the underlying electrical disease process and accommodate changing pacemaker/ICD needs,
- 4) re-programming around pacer related problems,
- 5) stay abreast of pacemaker advisories and alerts, and 6) educate patients and family members.
The following are key points to remember about this review of leadless pacemakers:
- The leadless pacemaker, which is 90% smaller than a transvenous pacemaker, is a self-contained generator and electrode system implanted directly into the right ventricle. The device is implanted via a femoral vein transcatheter approach; it requires no chest incision or subcutaneous generator pocket.
- The primary advantage of a leadless pacemaker is the elimination of several complications associated with transvenous pacemakers and leads: pocket infections, hematoma, lead dislodgment, and lead fracture. The leadless pacemaker also has cosmetic appeal because there is no chest incision or visible pacemaker pocket.
- Leadless pacemakers provide only single-chamber ventricular pacing and lack defibrillation capacity. Leadless pacemakers may be suitable for patients with permanent atrial fibrillation with bradycardia or bradycardia-tachycardia syndrome or those who infrequently require pacing. Leadless pacemakers are inappropriate for patients who require dual-chamber pacing, such as patients with certain forms of heart block or sinus node dysfunction.
- Only one leadless pacemaker (Micra [Medtronic PLC; Minneapolis, MN]) has been approved by the US Food and Drug Administration for use in the United States; a second (Nanostim [Abbott Laboratories; Abbott Park, IL]) is pending approval.
- Micra attaches to the right ventricle myocardium via four linear self-expanding nitinol tines. Nanostim attaches via an active screw-in helix and secondarily via three nitinol tines angled perpendicularly to the helix.
- Low-molecular weight heparin is administered preoperatively and during the procedure to prevent development of thrombosis.
- Complications may occur related to femoral vein access or need for device repositioning; there is moderate risk of cardiac perforation with subsequent pericardial effusion.
- Current leadless pacemakers are designed to be compatible with magnetic resonance imaging.
- Battery life is approximately 5-15 years, comparable to that of a transvenous pacemaker. At end of battery life, a leadless pacemaker can be turned off and a new leadless or traditional pacemaker implanted. A leadless pacemaker is theoretically retrievable, but there is only limited experience with retrieval. Leadless pacemakers are likely to become encapsulated in cardiac tissue, as are the pacing leads of traditional pacemakers.
- The future of leadless device technology is promising and might eventually lead to expanded pacing capabilities. One beneficial application for leadless devices may be postoperatively following transcatheter aortic valve replacement. According to one study, 28% of patients require pacemaker about 5 days after transcatheter aortic valve replacement.

